Innovation in super-resolution 3-D microscopy
Innovation in super-resolution 3-D microscopy
| Author: | Emil Venere |
|---|---|
| Magazine Section: | Innovate |
| College or School: | CoE |
| Article Type: | Issue Feature |
| Feature Intro: | New achievements in "nanoscope" research promise to reveal dynamics in live-cell and tissue specimens. |
Innovations in super-resolution microscopy allow researchers to resolve structural and molecular features far smaller than the wavelength of light, representing a powerful and versatile laboratory tool, says Fang Huang, assistant professor of biomedical engineering at Purdue University.
The work promises capabilities such as deeper imaging of tissue samples and live cells, higher resolution of single molecules, and, eventually, animal-based studies.
“The wave nature of light restricts the resolution of conventional light microscopy to about 200 nanometers laterally and 500-700 nanometers in the depth direction, making details of subcellular structures and protein assemblies unresolvable,” Huang says.

(Photo supplied)
Ultra-high resolution imaging of whole cells
Huang is leading work to develop a next-generation “nanoscope” that promises to reveal dynamic mechanisms inside living cells in unprecedented detail. Molecules inside cells and in structures called organelles can be tagged with either “photo-switchable” fluorescent proteins or organic dyes that are able to glow when exposed to a small amount of ultraviolet light.
Super-resolution fluorescence microscopy earned its developers the Nobel Prize in Chemistry 2014 and has become an important tool in cell biology research. However, application of this technology has been limited because of challenges in resolving features deep below the surface of samples while maintaining the superior resolution in all three dimensions.
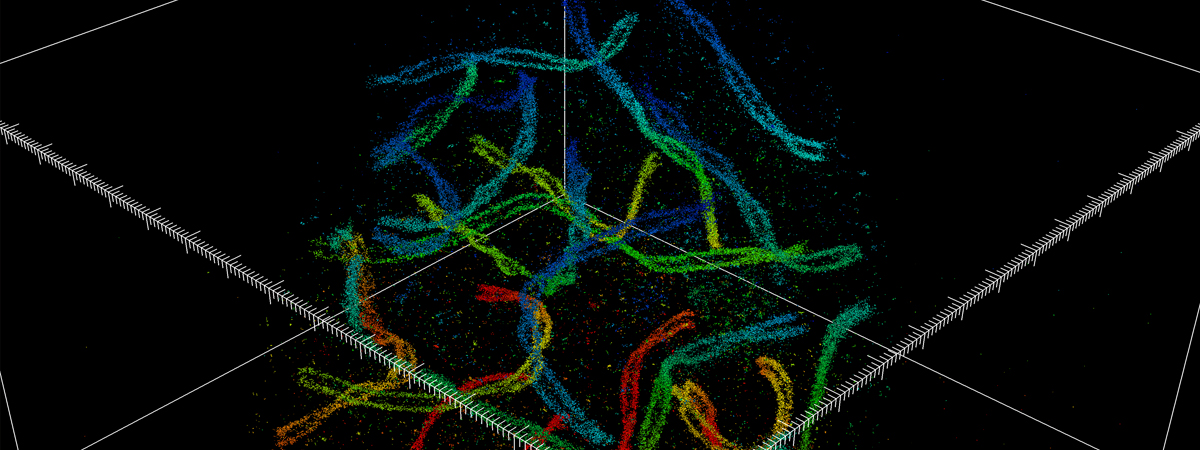
“We have now solved that problem with the development of the new nanoscope, a system called whole-cell 4Pi single-molecule switching nanoscopy (W-4PiSMSN). The approach exploits the wave nature of light by combining every emitted photon from single molecules through two independent but nearly identical optical paths in order to form an interference pattern,” Huang says. “Together with adaptive optics and novel algorithms, precise coordinates of each individual molecule can be derived from its interference patterns even when it is from an organelle deep inside the specimen.” Pinpointing the coordinates of thousands or millions of molecules makes it possible to reconstruct a 3-D image.
Huang's and colleagues’ research findings were detailed in a 2016 paper published in the journal Cell. The paper’s co-lead authors were Huang and Yale University postdoctoral research associate George Sirinakis. The team demonstrated the technology by imaging components in live cells, including synaptonemal complexes, which form links between homologous chromosomes; mitochondria and the endoplasmic reticulum, which are critical for cellular functions; the primary cilium, which protrudes from the cell as a sensory antenna; and bacteriophages, which are viruses about 50 nanometers in diameter that infect and kill bacteria.
The nanoscope system allows 3-D imaging of cellular constituents at 10- to 20-nanometer resolution throughout entire mammalian cells, powerful enough to reconstruct the fine features of viruses. Until now, such resolution was only possible using electron microscopy, which requires samples to be treated, killing the cells.

(Image courtesy of Huang et al./Cell)
Federal funding for super-resolution imaging
The Huang lab recently received two new federal grants: a $1.8 million award from the National Institutes of Health (NIH) and $500,000 from the Defense Advanced Research Projects Agency (DARPA).
“The NIH and DARPA awards facilitate two different research directions in my lab that also are synergistic parts,” Huang says. “I’m hoping we can combine elements of both projects to further push the envelope of super-resolution imaging and to shed light on biological processes that are otherwise hidden under the diffraction limit.”
The NIH Maximizing Investigators’ Research Award (MIRA) for New and Early Stage Investigators is a five-year grant for a project titled “Interferometric 3-D super-resolution imaging and structure and stoichiometry mapping in live cells.”
The DARPA Young Faculty Award will support two years of research with the potential for an additional year to develop a novel nanoscope that significantly improves imaging depth and volume while minimizing light-induced toxicity. The long-range goal is to allow super-resolution studies in live research animals.
Astronomy’s adaptive optics
To achieve this, Huang has harnessed the same kind of adaptive optics used in astronomy — deformable mirrors that change shape to compensate for light distortion. In astronomy, the deformable mirrors in telescopes are used to compensate for atmospheric distortion to yield clear images of celestial objects. Deformable mirrors also can be used to counteract the distortion caused when light passes through biological tissue.
Huang’s system integrates the deformable mirrors in a microscope using two objectives, one above and one below the sample, and introduces a set of new algorithms to pinpoint molecular positions of proteins deep inside cells.
“Originating from a molecular tag on cellular organelles, photons travel through two independent but closely matched interference arms, thanks to adaptive optics, then interfere with themselves to create a standing wave pattern that is extremely sensitive toward the exact position of the single molecule in the cell,” he says. “This sensitivity translates into a significantly improved resolution compared with conventional super-resolution methods.”
“We are excited to apply our imaging systems to study molecular mechanisms of cell division,” he adds. “Also we want to tackle other fundamental biological questions that have profound impact on our understanding of the nature of life.”
Comments